Science Corp has secured $230 million in fresh financing to accelerate commercialization of its first neural interface, positioning the company to move faster than many better-known brain-computer interface rivals. Led by Max Hodak, the co-founder and former president of Neuralink, the startup says it is closing in on market entry with a vision-restoring retinal implant called PRIMA.
A person familiar with the deal puts Science Corp’s post-money valuation at $1.5 billion. The round includes Lightspeed Venture Partners, Khosla Ventures, Y Combinator, Quiet Capital, and IQT, the strategic investor that supports technologies of interest to government agencies. With this raise, the company’s total funding reaches $490 million, and its headcount stands at about 150.
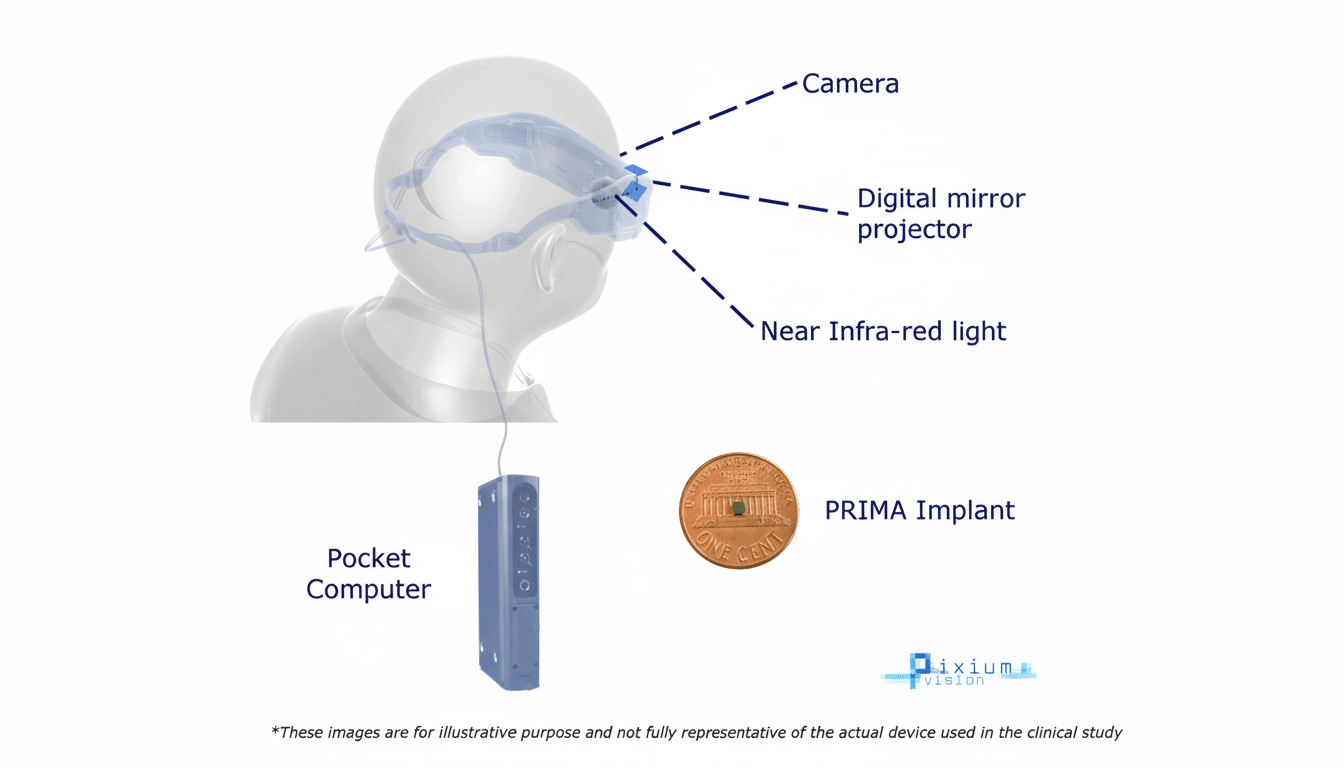
Inside the PRIMA retinal implant design and function
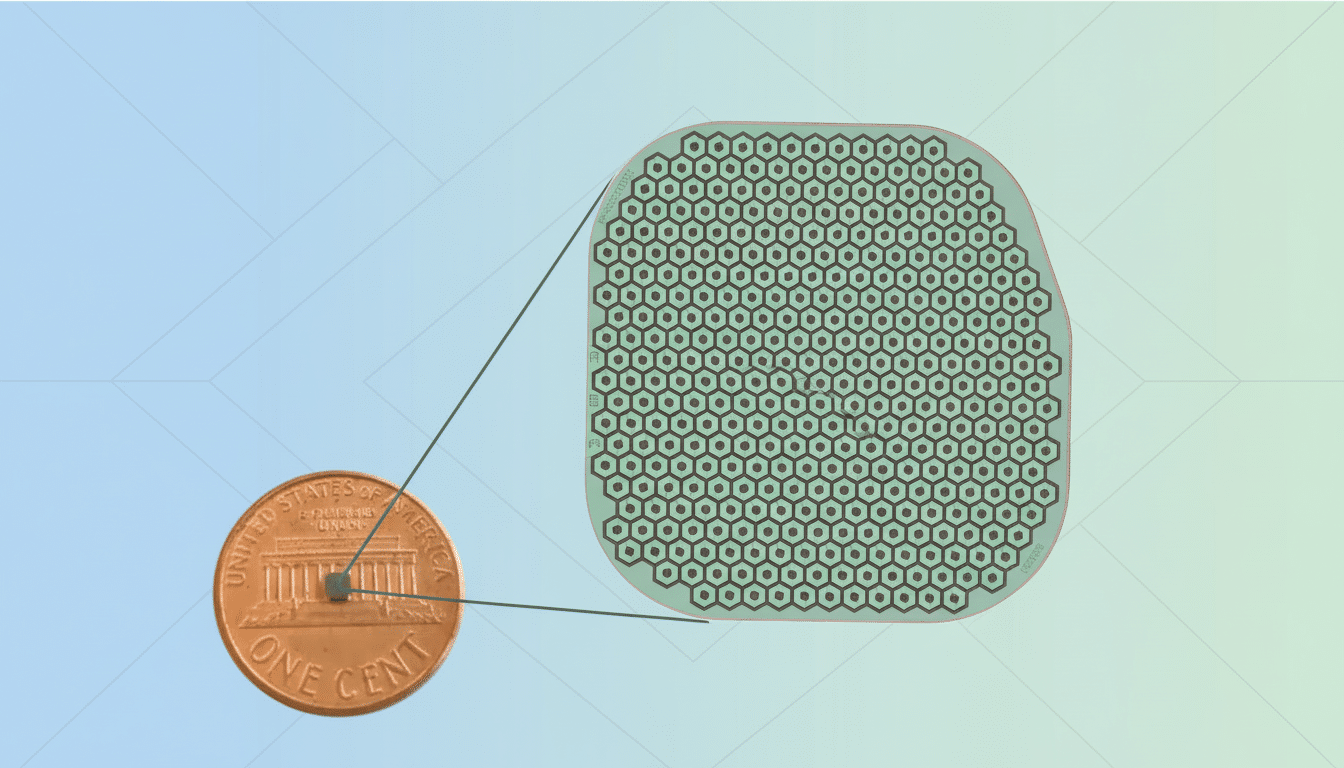
PRIMA is a photovoltaic chip—smaller than a grain of rice—designed to sit under the retina. Working in tandem with camera-equipped glasses, it converts images into light patterns that stimulate surviving retinal cells, routing visual information through the optic nerve to the brain. The approach aims to restore functional central vision for people with advanced age-related macular degeneration (AMD), where photoreceptors have been lost but the downstream visual pathway remains intact.
Science Corp acquired PRIMA from France-based Pixium Vision, then refined the implant and its external optics and completed trials that were already underway. Unlike intracranial BCIs that require a craniotomy, PRIMA leverages established subretinal surgical techniques familiar to vitreoretinal surgeons, potentially lowering procedural barriers and broadening the pool of implant centers.
Clinical results and regulatory path to market approval
Across trials involving 47 participants in Europe and the U.S., Science Corp reports that 80% achieved meaningful gains in visual acuity, with many able to read letters, numbers, and words after implantation. While the company has not publicly detailed all endpoints, the emphasis is on functional vision—tasks like character recognition and improved contrast sensitivity—rather than perfect natural vision.
The company has submitted a CE mark application under the European Union’s Medical Device Regulation and plans to prioritize a European launch once cleared. If successful, Science Corp contends it will be the first BCI company to put an implantable neural interface into commercial use. A subsequent U.S. rollout would likely navigate the FDA’s premarket approval pathway for active implantable devices.
The addressable need is considerable. AMD affects well over 190 million people globally, according to multiple epidemiological studies cited by vision research groups. Recently approved complement inhibitors in the U.S. can slow progression of geographic atrophy, but they do not restore lost central vision—leaving a clinical gap a prosthetic implant could fill.
Where it fits in the BCI race and competitive landscape
Science Corp’s ophthalmic route distinguishes it in a field dominated by motor and communication BCIs. Neuralink, Synchron, Blackrock Neurotech, and Precision Neuroscience are advancing implants intended to decode intent for cursor control or assistive typing. Those programs are in early human studies and have not reached broad commercial availability. By targeting the retina—part of the central nervous system but surgically accessible—Science may sidestep some adoption and safety hurdles that accompany cortical implants.
The company’s claim to be first will turn on regulatory timing, manufacturing readiness, surgical training, and reimbursement. Even with a green light, scaling sterile implant fabrication, calibrating external wearables, and standardizing patient selection criteria will determine how quickly clinics can deliver consistent outcomes.
Beyond PRIMA: new programs and emerging applications
New capital will also fund Science Corp’s biohybrid neural interface platform, an ambitious effort to grow engineered neurons from stem cells onto a waffle-like substrate that rests on the brain’s surface and forms synaptic connections with native circuits. If it works as intended, the approach could offer higher signal fidelity and long-term stability compared with purely electronic arrays—though it raises complex safety and regulatory questions that will require rigorous, staged studies.
The company has created a separate line of business called Vessel, focused on miniaturized organ perfusion. The goal is to enable organ transport on commercial flights and even home-based maintenance prior to transplant, a direction aligned with broader attempts in transplantation medicine to reduce ischemic injury and expand the donor pool. IQT’s participation underscores potential dual-use interest where resilient bio-infrastructure intersects with national capabilities.
What to watch next as Science Corp approaches launch
Science Corp is expanding its PRIMA trials to Stargardt disease and retinitis pigmentosa, two inherited retinal disorders that often strike younger patients. Positive outcomes there would widen the clinical footprint beyond AMD and bolster the case for payer coverage.
The sprint from clinical data to real-world deployment will hinge on three things:
- Reproducibility of visual gains outside controlled trials
- Surgeon training that keeps complication rates low
- Pricing that reflects value in restored independence
If Science Corp clears these hurdles, it won’t just notch a win in the BCI race—it could redefine what practical neurotechnology looks like for patients.

